 NITRILE GLOVES
NITRILE GLOVES
Introduction
Natural rubber (NR) latex has been used as the main ingredient to produce medical gloves and industrial gloves for a long time. This is because the material is easy to use by the manufacturers. In addition, the products have excellent performance properties such as tear strength, tensile strength, fit and comfort. However, due to the risks of Type I allergy caused by certain residual NR proteins in the gloves, the use of synthetic rubber gloves has been growing steadily over the past 25 years, particularly in the healthcare settings. Currently, there are several synthetic latexes or dispersion that are used in the manufacturing of medical gloves. The major ones include carboxylated acrylonitrile butadiene copolymer (also known as nitrile), polychloroprene, polyvinyl chloride (also known as PVC or vinyl rubber) and synthetic polyisoprene. Of these, nitrile latex is the most popular material, particularly for examination glove applications due to its relatively low cost compared to polychloroprene and polyisoprene latexes, with reasonably good-performance properties such as tensile strength and rounded probe puncture resistance. Although PVC is cheaper than nitrile, it normally contains plasticiser which could be of health concern to users. Moreover, PVC gloves are known to be less durable during use. Therefore, the requirements of ASTM D 5250 on tensile strength and elongation at break for PVC examination gloves of at least 11 MPa and 300%, respectively, are much lower than nitrile examination gloves of at least 14MPa and 500% as stated in ASTM D 6319. Natural rubber examination gloves, on the other hand, are expected to have at least 18 MPa and 650% for tensile strength and elongation at break, respectively as specified in ASTM D 3578, the highest among the three materials. This article describes some of the common properties of carboxylated nitrile latex and nitrile examination gloves, including the processing of nitrile latex into gloves.
Properties of Carboxylated Nitrile Latex
Carboxylated nitrile latex is prepared by polymerising three types of monomer using the emulsion polymerisation technique. The three monomers are butadiene, acrylonitrile, and acrylic acid. Table 1 shows a typical recipe of emulsion polymerisation to produce carboxylated nitrile latex.
| Component | Parts by weight |
|---|---|
| Butadiene (gas) [Monomer] | 70 |
| Acrylonitrile (liquid) [Monomer] | 25 |
| Acrylic acid (liquid) [Monomer] | 5 |
| SDBS Anionic Surfactant | 4.5 |
| Water | 180 |
| Initiator | 0.5 |
| Tetrasodium Pyrophosphate (Buffer) | 0.1 |
| Sodium chloride | 0.3 |
| Reaction conditions | 30℃/24hrs |
| Polymerization yield | 90% |
-
Reference:
- Sugimura T., Kataoka Y., Suzuki S., and Kasahara K. “Application of Synthetic Latex” 1993; published by Society of Polymer, Japan
Upon completion of the polymerisation, surfactant, defoamer, antioxidant and biocide are normally added to the latex. The latex has a smaller particle size and narrower particle size distribution than natural rubber latex with a median value of about 0.1 micron in diameter. The pH is normally in the range of 8.0 to 8.5. The latex also has a high gel content, indicating the occurrence of branching and crosslinking during the polymerisation process. The pressure stability of nitrile latex is lower compared to that of natural rubber latex, which therefore requires more delicate handling. Due to the high surfactant level needed, the latex is foamier than NR latex. Like other commercial synthetic latexes, the shelf life of nitrile latex is usually six months whereas NR latex can be used even after being stored for a year. Typical properties of commercial carboxylated nitrile latexes, as given by the suppliers, are shown in Table 2.
| Latex Product Code | Coagulum (%) | TSC (%) | pH | Viscosity (mPa.s) | Surface tension (mN/m) | Particle size (nm) | T(g)(℃) |
|---|---|---|---|---|---|---|---|
| LX550L | - | 45 | 8.3 | 60 | 35 | 110 | -27 |
| X6311 | - | 45 | 8.2 | 50 | 31 | - | - |
| L105 | 0.02 | 45 | 8.4 | 100max | 33 | 122 | - |
| Note: Some of the properties were not given by the respective suppliers | |||||||
Properties of Carboxylated Nitrile Rubber
Butadiene is the soft component of nitrile rubber. It is non-polar, providing the elastic properties to rubber. It also functions as a reaction site for sulphur vulcanisation of rubber. Acrylonitrile and acrylic acid, on the other hand, are the hard component giving stiffening effects and strength to rubber. Acrylonitrile is responsible for the oil-resistance properties while acrylic acid is for tensile strength, tear resistance and abrasion resistance. Carboxylic acid group from acrylic acid can be a crosslinking site for metal ions such as zinc, magnesium and aluminium. Because of this, carboxylated nitrile rubber has better mechanical properties than non-carboxylated nitrile rubber. The different grades of commercial nitrile latex are normally determined by their acrylonitrile contents. A high acrylonitrile content will increase the solvent resistance, glass transition temperature, gas impermeability, stiffness, abrasion and ageing resistance of rubber. Unlike natural rubber, carboxylated nitrile rubber does not crystallise upon stretching.
Manufacturing of Nitrile Glove
a) CompoundingLike natural rubber latex, it is necessary to vulcanise carboxylated nitrile latex to enhance the strength of nitrile gloves. The vulcanisation is carried out by adding compounding chemicals to the latex before being processed into gloves. The compounding chemicals consist of stabilisers, protecting agents such as antioxidant and antiozonant, vulcanising agents such as sulphur, zinc oxide and accelerator. In the vulcanisation of natural rubber latex, zinc oxide functions as an activator but in the case of carboxylated nitrile latex, it also serves as a curing agent to form ionic crosslinks with the carboxylic groups of rubber. The mixture is then allowed to mature for a period of time before being used in the production line.
The online dipping process is summarised in Figure 2 below.
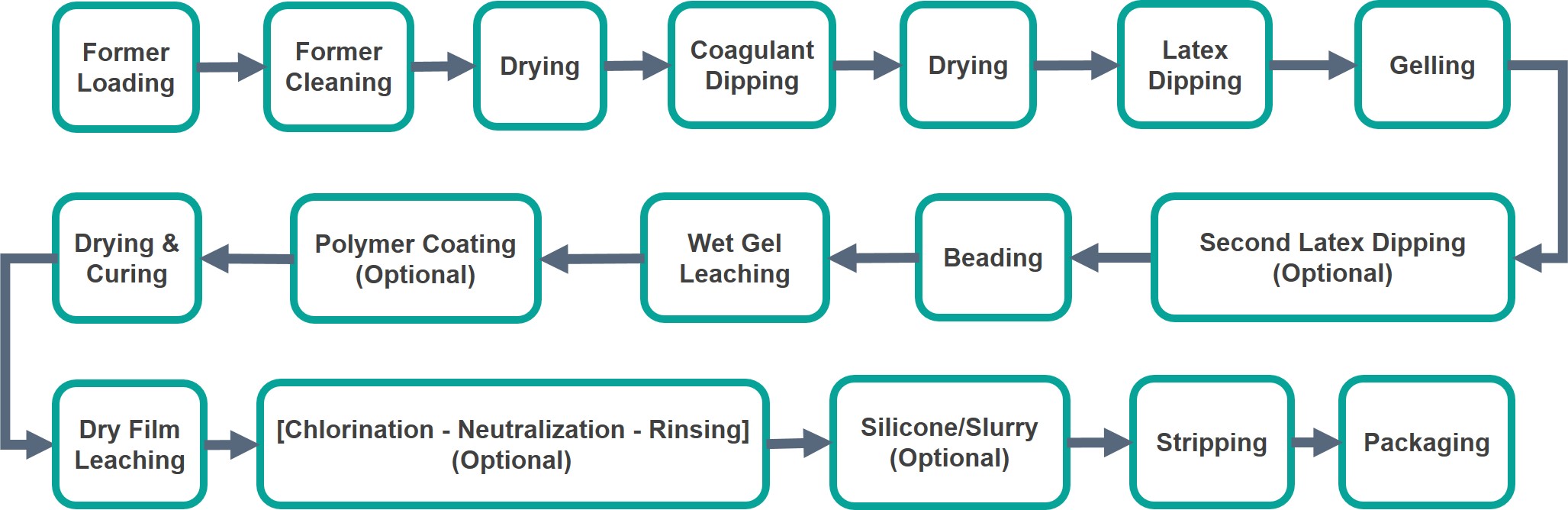
The former holder may consist of single, double or batten with multiple formers. Depending on the line size, normally 2 - 3 sizes are loaded per dipping line for examination gloves and more sizes for longer production lines.
For powdered coagulant, nitric acid and sodium hypochlorite are used while powder-free coagulant normally involves the use of commercial cleaning agent supplied together with powder-free coagulant which normally contains stearate compounds.
The main ingredient of coagulant is normally calcium nitrate. Other minor components include wetting agent, powder for mould release purpose such as calcium carbonate or stearates, stabiliser, defoamer, and dewebber. In some cases, thickener is also used to improve the coagulant pickup especially for surgical gloves.
In most cases, the coagulant is prepared by dissolving salts, such as calcium nitrate in water but alcohol can also be used. The product mould is then dipped in the coagulant to form a thin layer. This is normally used to produce unsupported rubber gloves. The thickness depends on many factors such as coagulant concentration, viscosity of latex, dwell time, withdrawal rate and chemical stability of latex.
Carboxylated nitrile latex is able to form a thinner film than natural rubber latex. To enhance quality, a second latex dipping is normally adopted in the manufacturing of nitrile gloves. This double-latex dip has also been found to improve the tensile properties of the final products, particularly for chlorinated nitrile gloves as shown in Table 3.
| Conditions | Unaged Tensile Strength, MPa | Aged Tensile Strength, MPa |
|---|---|---|
| Single Dip 1 (Chlorinated) | 30.4 | 30.7 |
| Double Dip 1 (Chlorinated) | 41.4 | 41.2 |
| Single Dip 2 (Non-Chlorinated) | 41.4 | 37.5 |
| Double Dip 2 (Non-Chlorinated) | 42.0 | 38.9 |
-
Reference:
- Ng T.H., Tong H.K., Lum L., Sayuti M.M., Khoo N. “Nitrile Glove Performance: New Dimensions in Analysis and Improvement” Proc Int. Latex Conf. Akron 2015; Paper 12.
The thin latex film on each former is allowed to fully gel and leached in hot water at 50 – 70℃ for 1 - 5 minutes to remove water-soluble materials such as surfactant, and calcium salt. Wet-gel leaching can help to integrate the latex film and improve mechanical properties such as tensile strength as shown in Table 4. It can also reduce the odour of products. The temperature is normally kept below 70℃ as high temperatures may cause the weak wet gel to crack.
| Conditions | Unaged Tensile Strength, MPa | Aged Tensile Strength, MPa |
|---|---|---|
| Control (Unleached) | 27.9 | 30.4 |
| Wet gel leached | 41.4 | 41.2 |
-
Reference:
- Ng T.H., Tong H.K., Lum L., Sayuti M.M., Khoo N. “Nitrile Glove Performance: New Dimensions in Analysis and Improvement” Proc Int. Latex Conf. Akron 2015; Paper 12.
For the manufacturing of powdered gloves, the gloves are dipped into corn-starch powder slurry to pick up a coat of lubricant that makes them easier to strip and don when used. They are then further dried.
For powder-free gloves, a step of silicone dip may be introduced to facilitate stripping and donning by the users. Nowadays, most of the ex-machine nitrile gloves are powder-free. In the case of cleanroom gloves, the use of silicone is prohibited.
Except for cleanroom nitrile gloves, most of the thin nitrile gloves such as those for medical examination applications, do not require offline process. This is because powder-free nitrile gloves can be easily produced by the online process. Moreover, due to the permanent set properties, thin nitrile gloves such as those for medical examination applications could become creased when processed offline in bulk. However, as nitrile gloves for cleanroom applications are normally chlorinated on the inside and outside surfaces and washed repeatedly offline to reduce extractable materials, the additional offline process is necessary.
Advantages of Nitrile Gloves
Nitrile gloves have been reported to be suitable for medical applications in terms of their barrier properties. In addition, there are many advantages of producing or using nitrile gloves. Some of these are described here.
-
Reference:
- Edlich R.F., Suber F., Neal J.G., Jackson E.M., Williams F.M. “Integrity of Powder-Free Examination Gloves to Bacteriophage Penetration” J. Biomed Mater. Res. (Appl Biomater) 1999; 48, 755–758.
- Landers T.F., Dent A. “Nitrile versus Latex for Glove Juice Sampling” PLoS ONE 2014; 9, e110686.
- Korniewicz D.M., Maher M. El-Masri M.M., Broyles J.M., Martin C.D., O’Connell K.P. “A Laboratory based Study to Assess the Performance of Surgical Gloves” AORN J., 2003; 77, 772-779.
- No proteins
As mentioned in Section 1, nitrile gloves contain no proteins. They are therefore suitable for use particularly by healthcare workers who are allergic to NR proteins.
- Thinner gloves
Nitrile gloves can be made at a lower thickness than NR gloves which provide a better sensitivity to glove users. The raw material consumption is also lower than NR gloves.
- Puncture resistance
Nitrile gloves are also known to have a better puncture resistance against a rounded probe than NR gloves. However, it must be noted that when tested against a sharp needle, both gloves have a similar puncture resistance as shown in Table 5.
- Oil resistance
Nitrile rubbers are polar materials and therefore their resistance against polar liquids such as oils, toluene, petrol, is better than NR or synthetic polyisoprene which are non-polar materials.
- Antistatic
The higher polarity of nitrile gloves also gives them the advantage of having antistatic properties which make the gloves suitable for handling semi-conductor products. Because of this, most of the cleanroom gloves in the market are made from carboxylated nitrile latex.
- Accelerator-free
Due to the presence of carboxylic acid groups which can be used as a crosslinking site, it is easier to produce accelerator-free nitrile gloves than accelerator-free NR gloves. This will address the Type IV allergy issue faced by some sensitive individuals. Some examples of accelerator-free nitrile gloves can be found in US Patents 9279038, 9243117, and 8399105.
- Ozone resistance
The properties of stress relaxation of nitrile gloves make them less prone to ozone attack when compared to NR gloves. Therefore, ozone cracking rarely occurs in nitrile gloves during storage.
| Puncture force, N | ||
|---|---|---|
| Probe (ASTM F 1342)* | 0.4mm needle** | |
| NR Latex | 3.4 | 0.036 |
| Nitrile 1 | 8.6 | 0.024 |
| Nitrile 2 | 9.5 | 0.025 |
| Nitrile 3 | 11.3 | 0.033 |
| *2mm Probe; **Similar to ASTM F 2878, 28G needle | ||
-
Reference:
- Patel H.B., Fleming G.J.P. and Burke F.J.T. “Puncture resistance and stiffness of nitrile and latex dental examination gloves” Brit. Dent. J. 2004; 196, 695–700
Opportunity for Improvement
Several properties of nitrile gloves can be further improved to enhance product performance and expand applications.
- Stiffness
Nitrile gloves are stiffer than NR gloves. Therefore, the fit and feel properties are not as good as NR gloves especially when used for a long period of time.
- Peter Mylon P., Lewis R., Carré J. Martin N., Brown S. “A study of clinicians’ views on medical gloves and their effect on manual performance” Am. J. Infect. Control 2014; 42, 48-54
- Stress relaxation
Unlike NR gloves, nitrile gloves take a longer time to return to their original shape when the stretching force is released. For surgical glove applications, the cuff is required to grip instantly to the sleeve of surgical gown upon donning. However, for examination glove applications, such requirement is not needed.
- Meeting standards
Due to different requirements of ASTM 6319 and EN 455 series for medical examination applications, it is difficult to meet both standards with just one nitrile glove. The minimum elongation at break of 500% for ASTM standard would render the glove too soft to meet the EN 455 series requirement of minimum 6N force at break. Manufacturers normally produce 2 variants of nitrile gloves for the US and EU markets. NR gloves, on the other hand, can meet both standards with just one glove.
- Polar liquids
Being a polar material, the resistance of nitrile gloves against polar liquids such as alcohol, and acetone is not as good as NR gloves.
- Tear resistance
Tear resistance of nitrile gloves is not as good as that of NR gloves
-
Reference:
Malaysia’s Strengths
Based on more than a century of experience in producing NR and NR products, particularly NR gloves, the Malaysian rubber industry has successfully applied skilful technical know-how on the production of NR products to synthetic products such as nitrile gloves. With the benefits of experience and technical know-how, Malaysia is now the world’s largest producer and exporter of quality nitrile gloves. Together with NR gloves, Malaysia has captured a 64% global market share of rubber gloves. Furthermore, the strong competition among the glove manufacturers has led to production efficiency improving tremendously via automation and innovation. The production speed leapfrogged from just 3000 gloves per hour in 1988 to 45,000 per hour in 2013. The number of workers required to produce one million pieces of gloves has also reduced from 9.7 in 2009 to 3.6 in 2016. At the same time, product quality has also improved tremendously to AQL 0.65 or even lower. The competitive manufacturing environment in Malaysia also drives the commercialisation of new products and product portfolio diversification such as accelerator-free nitrile gloves and damp-hand donnable nitrile gloves. To further improve production efficiency, the manufacturers have also started to embrace industry 4.0, which involves the integration of robotics, real-time data collection via remote-controlled sensors, digitisation, analytics, and integration of different production facilities via internet for easy management, cost reduction, quality control and maintenance. Growth of the rubber glove industry in Malaysia has also led to the establishment of related eco-systems such as machine fabricators, chemical suppliers, and synthetic latex manufacturers. Supporting the industry is the Malaysian Rubber Export Promotion Council which actively promotes rubber gloves in international markets thereby enhancing market visibility of Malaysian rubber gloves.
-
Reference:
- Low J.F. “MARGMA Industry Brief on the Rubber Glove Industry” A paper presented at One World Hotel, 15 July 2016
Concluding Remarks
Although the issue of Type I allergy due to the proteins in NR gloves has decreased in recent years, the use of disposable synthetic gloves to replace NR gloves, particularly in the examination glove segment as part of risk management, has been growing significantly over the past 25 years. Nitrile gloves are a good synthetic alternative due to its comparatively low cost with good-performance properties such as barrier, puncture resistance, and oil resistance when compared to polychloroprene and polyisoprene gloves. They can easily be made thinner and hence reduce material consumption. Accelerator-free system can be adopted easily for manufacturing nitrile gloves which also helps to address the Type IV allergy issue, in addition to the Type I allergy issue. However, nitrile gloves are generally stiffer than NR gloves and the stress-relaxation properties limit the use of such gloves for surgical applications. More research is thus needed to enhance their flexibility and elasticity in order to expand the applications of nitrile gloves.