 HEALTH SAFETY
HEALTH SAFETY
The Centers for Disease Control and Prevention (CDC) estimates that 2 million people pick up infections during hospital stays each year, resulting in about 90,000 deaths with an overall cost of $5 billion annually. For this reason, effective protection is critical to both healthcare professionals and their patients. Both the National Institute for Occupational Safety and Health (NIOSH) and the Occupational Safety and Health Administration (OSHA) recommend the use of appropriate barrier protection when handling blood pathogens and infectious materials. The use of medical gloves with effective barrier performance provides one such safety measure. In view of the availability of many types of medical gloves today, and that not all gloves offer the same degree of barrier protection, it is important that care must be taken to select the right gloves for the right procedures.
- Hospital Infection Rate
Barrier Protection
In the healthcare environment, disposable medical gloves are life-saving devices that prevent the transmission of AIDS, hepatitis B and other potentially dangerous infectious diseases that can be contracted through contact with blood and body fluids. Effective barrier protection should therefore be an important criterion for glove selection and glove use.
Considerable information is now available as shown by a great number of comparative studies on barrier performance of the various types of medical gloves by U.S. researchers (Please see Table 1). Findings invariably showed that non-latex (synthetic) gloves, especially those made of vinyl, have inferior barrier with significantly higher leakage rates during use as compared to natural rubber latex gloves. In fact, natural rubber latex gloves have been universally preferred because of their excellent barrier protection, in-use strength, comfort, fit, and tactile sensitivity - important characteristics of gloves, as failure of these could lead to barrier protection being compromised. Furthermore, natural rubber latex gloves have also resealing properties when tiny needle punctures are encountered, a feature not observed in synthetic gloves such as vinyl and nitrile gloves.
"NRL (natural rubber latex) is pliable allowing for natural molding for more appropriate fit and has the ability to reseal when tiny punctures occur. In general, NRL provides comfort to the wearer, adequately protects against microorganisms, and provides adequate barrier effectiveness when used for medical and nursing procedures. Consequently, NRL is still the barrier of choice in the U.S."
Resealing Properties
- Reference:
- Korniewicz, D.M., Laughon, B.E., Cyr, W.H., Lytle, C.D., and Larson, E. "Leakage of Virus through Used Vinyl and Latex Examination Gloves", Journal of Clinical Microbiology, 1990; 28: 787- 788.
- Korniewicz, D.M., El-Masri, M., Broyles, J.M., Marin, D.C. and O'Connell, K.P "Performance of latex and non-latex medical examination gloves during simulated use." Am. J. Infect. Control, 2002; 30: 133-138.
- Korniewicz, D.M., Garzon, L. and Plitcha, S "Health Care Workers: Risk Factors for Nonlatex and Latex Gloves During Surgery." Journal of American Industrial Hygiene Association (AIHA), 2003, 64:851-855.
- Korniewicz, D.M., Seltzer, J. and Feinieb, M. AM. "Failure Rates in Nonlatex Surgical Gloves." J. Infect. Control, 2004, 32: 268-273.
- Klein, R.C., Party, E. and Gershey, E.L. "Virus Penetration of Examination Gloves." Biotechniques,;1990; 9: 196.199.
- Olsen R.J, Lynch P, Coyle M.B., Cummings J, Bokete T. and Stamm W.E. "Examination Gloves as barriers to Hand Contamination in Clinical Practice" JAMA 1993; 270: 350-353.
- Douglas A, Simon T. and Goddard M. "Barrier Durability of Latex and Vinyl Medical Gloves in Clinical Settings" Am. Industrial Hygiene Association J, 1997; 58: 672-677.
- Rego A. and Roley L. "In-use Barrier Integrity of Gloves: Latex and Nitrile Superior to Vinyl." Am. J. Infection Control; 1999: 27(5): 405-410.
- Kerr L.N., Chap M.P., Cash L.D., O'Malley L.G., Sarhrani E.M., Teixeira J.C., Boivin W.S. and Mailhot S.A. (FDA 2004) "Assessment of th Durability of Medical Examination Gloves." J. Occupational & Environmental Hygiene, 2004; 1:607-612.
Resealing Properties
Natural rubber latex gloves allow for less viral penetration than vinyl and nitrile gloves due to their resiliency and "resealing" properties.
- Reference:
- ABSTRACT - Barrier Performance of NR, Vinyl and Nitrile Gloves on Punctured, Hasma, H. and Othman, A.B, Paper presented at the "Latex 2001" Conference, RAPRA, 4-5, December 2001, Munich, Germany. Also published in J. Natural Rubber Research, 2003; 6(4): 231-240.
- ABSTRACT - A PCR-Based Method for Detecting Viral Penetration of Medical Examination Gloves, Broyles J.M., O'Connell K. P. and Korniewicz D. M., J. Clin. Microbiol. 2002; 52: 965-999. Also reported in "Barrier integrity of medical gloves," paper presented by Korniewicz D. M at International Glove Conference, Malaysia, 2002.
Many studies highlight the superior barrier protection qualities of latex.
Journal of Arthroplasty, 2010
The perforation rates of latex-free surgical gloves and latex surgical gloves in a clinical trial during primary hip and knee arthroplasty were compared and analyzed. The overall latex glove perforation rate was 8.4% compared to 21.6% for latex-free alternatives (χ2 P< 0.001). The operation perforation rate for latex gloves was 34.4% compared with 80% for latex-free gloves (χ2 P< 0.001). The researchers suggested that the latex-free gloves tested could not provide a reliable barrier between the surgeon and the patient. As such, they questioned the safety of these gloves and the standards set by the regulators.
Journal of American Industrial Hygiene Association (AIHA), November/December 2003.
This study examined glove failure and related factors in both non-latex (neoprene and nitrile) and latex surgical gloves after routine use. A total of 11,118 usable surgical gloves were tested. The findings revealed that the nonlatex surgical gloves were significantly more likely to fail than latex gloves.
American Journal of Infection Control, 2004
This study compared the frequency of glove defects for non-latex (neoprene and nitrile) and latex surgical gloves while surgeons performed routine surgery, and to evaluate surgeons' satisfaction with nonlatex sterile gloves. A total of 6,386 gloves used by 101 surgeons and 164 residents representing 15 surgical services were included in the study. Results showed that while intact nonlatex and latex surgical gloves provide adequate barrier protection, nonlatex surgical gloves have higher after-use defect rates and lower user satisfaction than latex surgical gloves.
Journal of Occupational and Environmental Hygiene
This study determined the durability of various types of medical gloves using a laboratory test developed by researchers. Results were also compared with a simulated clinical method. Durability of powdered and powder-free vinyl gloves was shown to be inferior to that of natural rubber latex gloves and other types tested. The failure rates of vinyl ranged from 24% to 42%, compared to 3% - 17% for other types tested. Results were consistent with those shown by the simulated clinical method.
American Journal of Infection Control, August 2002
The study found nitrile examination gloves to be a suitable alternative to latex, whereas vinyl and copolymer examination gloves were found to be less effective barriers. The study concluded that further research would be needed to determine whether nitrile gloves can provide effective barrier qualities during clinical use versus laboratory simulations.
The study, presented at a conference by the Malaysian Rubber Board, found that the low leak rate from punctured natural rubber latex gloves was observed to be associated with their low modulus and high tear strength resistance compared to those of vinyl and nitrile gloves.
Dental Material, March 2000.
The study found that some natural rubber latex gloves showed a low rate of leakage, while vinyl and nitrile gloves were penetrated quickly and to a great extent.
American Journal of Infection Control, October 1999.
The study found that leakage rates ranged between 12% and 20% for stretch vinyl gloves and between 26% and 61% for standard vinyl gloves. Leakage rates for latex and nitrile gloves were only 0% to 4% and 1% to 3%, respectively.
American Industrial Hygiene Association Journal, September 1997
The study found that leakage rates ranged between 22% and 27% for stretch vinyl gloves and between 26% and 32% for standard vinyl gloves. A rate of only 1% was found for powder-free latex gloves.
American Journal of Infection Control, February 1994
The study found that vinyl gloves were found to leak significantly more when worn one at a time than when double gloves were used (51.3% and 19.7% respectively). However, there was no difference between the very low leakage rates for single and double latex gloves (4.1% and 3.8% respectively).
JAMA, the Journal of the American Medical Association, July 1993
The study found that under conditions of routine use, gloves effectively function as a protective barrier even when leaks are present. Latex gloves were less frequently associated with leaks and hand contamination.
American Industrial Hygiene Association Journal, January 1993
The study compared leakage rates of used latex and vinyl examination gloves from high and low risk clinical units. The researchers found that regardless of stress and duration worn, 85.3% of used vinyl gloves and 18.4% of used latex gloves leaked.
Biotechiques, August 1990
The study found that thin gloves manufactured from polyethylene or polyvinyl chloride are ineffective barriers while gloves of thin latex are superior. The study found that 22% of vinyl gloves and 40% of polyethylene gloves leaked virum (lambda phage) while leakage was detected in less than 1% of latex.
Journal of Clinical Microbiology, April 1990.
The study found that viral penetration in vinyl gloves (phiX 174) was 63% in contrast to only 7% for latex gloves.
Latex Allergy
The awareness of latex protein allergy in the 1990s affecting certain sensitive individuals had raised concern about the use of latex gloves. It is apprent now that the cause of the allergy arose from the use of an older generation of latex gloves that had no control on their residual protein level. Natural rubber latex gloves are derived from latex obtained from the Hevea Brasiliensis tree. Like all plant materials, latex contains proteins, which can cause sensitivity among certain sensitive individuals. The latex protein allergy is a type 1 reaction with immediate hypersensitivity. Sensitization is brought about by repeated exposures to excessive residual soluble protein in the latex gloves. As with other proteins in nuts, fruits, potatoes, tomatoes and some seafood. Which can elicit type 1 allergy, the reactions produce similar symptoms ranging from very mild common form of hives, watery eyes and runny noses, to asthma, to very serious but rare cases of anaphylaxis. Studies have shown that latex protein allergy affects about 1% of the general population (Liss G.M, and Sussman G.L. Am. J. Industrail Medicine; 1999; 35:196-200). The prevalence among healthcare workers, spina bifida and multiple-operated children is higher. Once sensitized and latex allergic, these individuals have to avoid the allergens they are sensitive to.
Latex protein allergy is sometimes confused with the more common irritant contact dermatitis and allergic contact dermatitis, the two other types of possible reactions often experienced by sensitive glove users. While irritant such as detergents, powder, temperature and pH extremes are causes of the former, the latter is mainly due to the presence of residual chemicals used in the manufacturing of gloves. Both latex and non-latex gloves can bring about these two types of reactions.
Latex Safe Measures
Advancement in glove manufacturing technologies, especially in Malaysia, has led to the production of a new generation of low-protein latex gloves, which remain effective in barrier protection and are low risk to latex protein allergy.
The use of such low-protein gloves has been shown by many studies by researchers in the U.S., Europe and Canada confirming that wearing low-protein low- or powder latex gloves greatly diminishes the risk of allergic reactions and the likelihood of healthcare workers developing latex sensitivity. These studies also demonstrated that latex sensitive individuals donning synthetic gloves could work safely alongside their co-workers wearing low-protein latex gloves.
- Hospital Studies
In fact, to assist users in identifying quality low-protein latex gloves, Malaysia has formulated the Standard Malaysian Glove certification program to ensure the manufacture of quality gloves that are low-protein. Please see "SMG" gloves.
Update on Latex Allergy
There has been a great deal of positive development regarding the decline of latex allergy since the 1990s, attributed to efforts made by the glove industry in product improvements and education by healthcare-related agencies. This has in fact been acknowledged recently by a number of well-known clinical researchers.
- Good News for People with Latex Allergies
Latex allergy prevalence is down since the mid-1990s, mainly due to a change in the way latex gloves are manufactured.
P. J. Early, HealthLink, Medical College of Wisconsin Aug 10, 2005 - Lessons Learned from Latex Allergy
Most latex-sensitized healthcare workers can today be accommodated in the 'new' latex allergen-safe environment. Low-protein, powder-free gloves have drastically reduced exposures in the healthcare setting.
Beezhold D.and Sussman G., Business Briefing: Global Surgery - Future Directions 2005 Sep 2005 - Latex Allergy in the Surgical Environment
"It appears that the epidemic has been eliminated."
Jordan N Fink Business Briefing: Global Surgery - Future Directions 2005 Sep 2005. - Latex Allergy has almost disappeared among healthcare workers.
Latex allergy has very significantly declined and almost disappeared. It is because of the initiatives of the government, hospitals and industry, which have changed their products. The examination gloves are 1,000-fold less allergenic and sterile gloves about 100-fold less allergenic (than they were in the 1980s.)
Sussman G.M., University of Toronto, Hospital Employee Health, AHC Media, LLC Website, 7 January 2007.
A Report on the Extractable Protein Content in Gloves under Lesen Mengeksport Sarung Tangan Getah (LMSTG) in 2018
Syuhaizat Md Ali, Hairul Ishyam Hussein & Dr Faridah Hanim Ab Hanan
Introduction
Restriction on the export of glove with protein content exceeding 400 μg/g is enforced by MRB under LMSTG to ensure only quality gloves meeting the requirement are exported from Malaysia. To ensure that the requirement is adhered to; the Malaysian Rubber Board (MRB) who is the authorized body to issue the LMSTG to glove exporters had carried out inspection and sampling of natural rubber examination glove (powdered) in order to monitor the level of extractable protein content.
Inspection and Sampling
A total of 215 inspections had been carried out to all license holders of LMSTG according to the type of business namely manufacturer, packer and trader and trading only. The types of businesses and the location of each license holders are shown in Graphs 1 and 2 respectively.
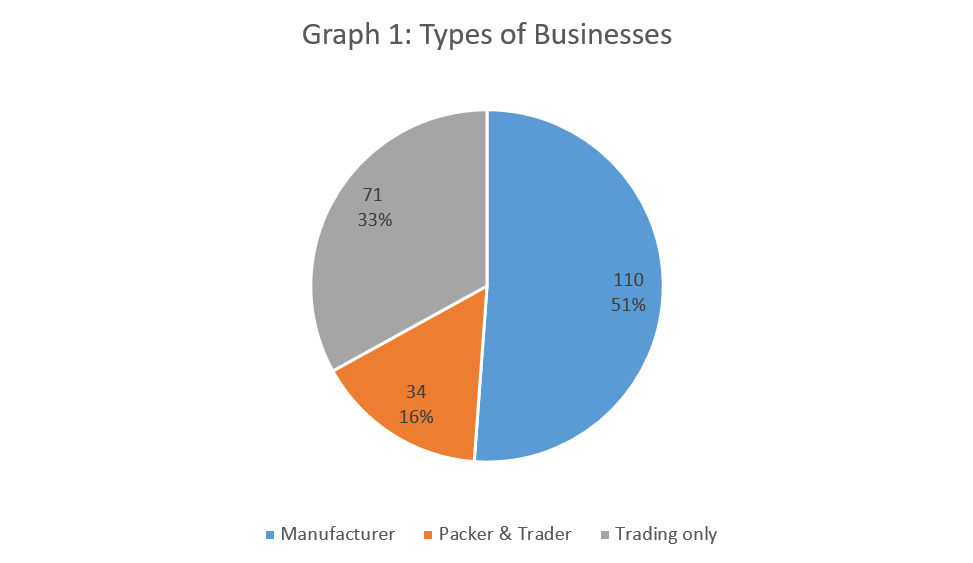
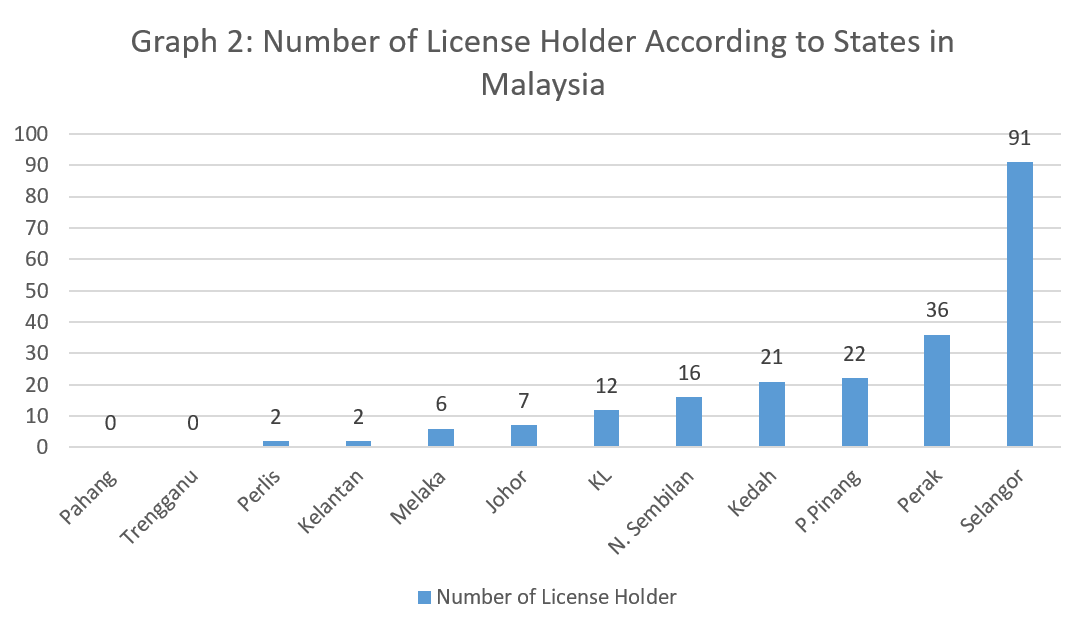
Based on the inspection carried out on all license holders, a total of 49 lots have been sampled. All samples were tested for extractable protein content (using ASTM D5712-2015 test method) at MRB testing laboratory which is accredited to ISO/IEC 17025.
Extractable Protein Content Result
The overall results have shown that all glove samples are meeting the specification of 400 μg/g. In term of year to year trend (2012 to 2018), the extractable protein content has shown a consistent decreasing trend (Graph 3) with the average value ranging from 256 μg/g in 2012 to 107 μg/g in 2018; a reduction of about 58% has been observed. This huge reduction has certainly indicated that the level of maturity of license holders in adopting the best industry practices, that emphasize on quality, has grown positively.
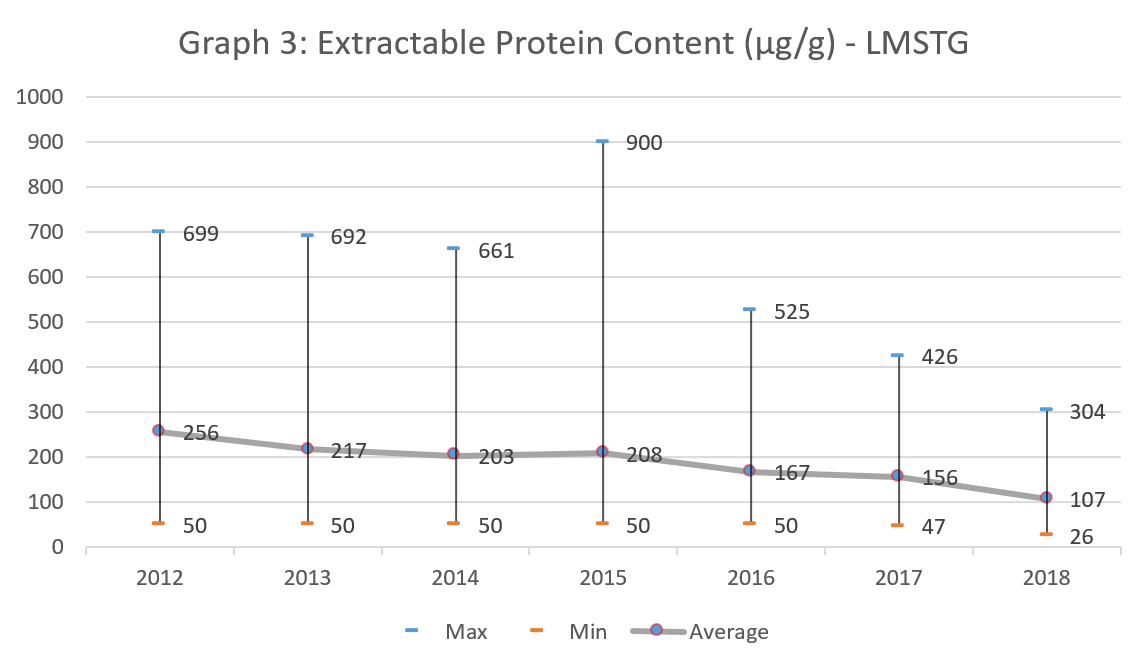
Conclusion
The amount of the extractable protein content in the gloves sampled for LMSTG has consistently decreasing since 2012 and this has demonstrated the technical capability in meeting the extractable protein content specification of 400 μg/g which is commendable.